Data-Driven Key Account Management for Clinical Research: Leveraging Free Trials and Competitor Insights
The clinical research landscape is undergoing a fundamental transformation as data-driven approaches revolutionize key account management (KAM). Where partnerships once relied primarily on personal relationships and intuition, today's successful clinical research organizations leverage real-time analytics, predictive insights, and comprehensive data integration to optimize their most valuable client relationships. This evolution from relationship-based to data-informed KAM strategies represents a critical competitive advantage in an industry where trial success rates, operational efficiency, and partnership longevity directly impact revenue and market position.
Practical Tip: Start your data-driven KAM journey by identifying 3-5 key metrics that matter most to your clinical research partners—whether it's patient enrollment rates, protocol adherence, or data quality scores. Track these consistently before implementing any new platform.
Free trials offer a strategic opportunity to evaluate data-driven KAM solutions without commitment. During your trial period, focus on these practical evaluation steps:
- Test data integration capabilities with your existing clinical trial management systems
- Assess real-time dashboard functionality for monitoring key partnership metrics
- Evaluate predictive analytics for identifying partnership risks and opportunities
- Validate data security and compliance features for sensitive clinical information
By leveraging free trials strategically, clinical research organizations can make informed decisions about implementing data-driven KAM solutions that deliver measurable ROI through enhanced partnership performance, reduced trial delays, and improved client retention.
Foundations of Data-Driven Key Account Management in Clinical Research
Effective data-driven key account management (KAM) in clinical research begins with precise account definition. Key accounts typically include strategic Contract Research Organizations (CROs), academic research institutions, and pharmaceutical partners with significant trial volume or specialized therapeutic expertise. These relationships transcend transactional vendor arrangements to become collaborative partnerships driving mutual success.
Essential data sources form the foundation of intelligent KAM. Track trial performance metrics including patient recruitment rates, protocol adherence, and timeline compliance. Monitor financial data like contract value expansion, renewal rates, and budget variance. Relationship indicators such as communication frequency, stakeholder engagement scores, and strategic alignment assessments provide qualitative insights. Practical tip: Create a partner scorecard with weighted metrics across these three categories for quarterly reviews.
Integrate data analytics into traditional frameworks through systematic implementation. Start by establishing structured data collection points at key study milestones. Use Clinical Trial Management Systems (CTMS) to automate KPI tracking and generate real-time dashboards. Implement monthly check-ins to review performance data and quarterly strategic business reviews for deeper analysis. Checklist: 1) Document RACI matrices for each partner, 2) Conduct needs assessments quarterly, 3) Establish feedback loops at study milestones, 4) Review and adjust strategies monthly based on performance data.
This data-driven approach transforms relationships from reactive problem-solving to proactive value creation, enabling CROs to anticipate sponsor needs, co-create solutions, and accelerate drug development while reducing costs.
Leveraging Free Trials for Strategic KAM Solution Evaluation
Free trials offer a unique opportunity to validate KAM solutions before committing. Start by identifying critical pain points: test how the platform addresses fragmented account data, manual reporting inefficiencies, and reactive relationship management. During the trial, assess data integration capabilities by connecting to your CTMS, EDC, and CRM systems—look for seamless API connections and real-time data synchronization.
Practical checklist for trial evaluation:
- Map 3-5 key clinical research accounts to test workflow compatibility
- Import actual trial data to validate analytics accuracy
- Simulate quarterly business reviews using platform dashboards
- Test cross-functional collaboration features with your team
- Measure time savings on manual reporting tasks
Evaluate user adoption by tracking how quickly your team adopts the platform—aim for 80%+ engagement within the first week. Assess workflow compatibility by running parallel processes: compare traditional methods against the KAM solution for account planning, stakeholder mapping, and performance tracking. Monitor real-time analytics capabilities by setting up alerts for trial milestones, budget variances, and relationship health scores.
💡 Tip: Create a scoring matrix (1-10) for each evaluation criterion and involve stakeholders from clinical operations, business development, and account management in the assessment. The most effective trials reveal not just technical capabilities but how the solution integrates with your existing clinical research ecosystem.
Key Metrics and Insights from Data-Driven KAM Implementation
Implementing data-driven Key Account Management (KAM) in clinical research transforms how you measure success. Track three core performance indicators: trial success rates (aim for >85% completion), timeline adherence (monitor deviations from projected milestones), and budget optimization (compare actual vs. planned spend with variance analysis). Use tools like RivalSense to benchmark against competitors.
For relationships, measure stakeholder engagement through communication frequency and response rates, communication effectiveness via feedback scores, and satisfaction with quarterly surveys. Set targets: >90% satisfaction scores and weekly check-ins with key contacts.
Business impact metrics include revenue growth from account expansions, partnership expansion (new sites or therapeutic areas), and competitive positioning—track your win rate against rivals in bids. Practical steps: 1) Establish a KAM dashboard with real-time data, 2) Conduct monthly reviews to adjust strategies, 3) Use insights to personalize outreach, and 4) Leverage competitive intelligence to anticipate market shifts. This approach ensures proactive management and sustainable growth.
Overcoming Implementation Challenges in Clinical Research KAM
Implementing data-driven Key Account Management in clinical research faces three major challenges. These obstacles often stem from organizational silos, resistance to change, and stringent regulatory requirements. First, data silos plague complex ecosystems where EHRs, EDC systems, CTMS platforms, and lab databases operate independently. Practical solution: Create a data integration checklist: 1) Map all data sources and formats, 2) Implement FHIR/HL7 standards for interoperability, 3) Use API-first platforms that connect disparate systems, 4) Establish a single source of truth dashboard.
Second, change management requires strategic planning. Start with a pilot program involving early adopters from clinical operations and account management teams. Create a phased rollout plan with clear milestones. Provide hands-on training sessions and establish super-users as internal champions. Measure adoption through usage metrics and feedback loops.
Third, regulatory compliance demands proactive measures. Ensure your KAM platform meets 21 CFR Part 11, GDPR, and HIPAA requirements. Implement role-based access controls, audit trails, and data encryption. Conduct regular security assessments and maintain detailed documentation for regulatory inspections. Partner with vendors who provide compliance certifications and transparent data handling policies.
Future Trends and Strategic Recommendations
Looking ahead, AI and predictive analytics will revolutionize key account management in clinical research. Emerging technologies are set to transform how we identify, engage, and retain key partners. By analyzing historical trial data, site performance metrics, and investigator profiles, AI can identify high-potential accounts before they're even on your radar. For example, predictive models can flag sites with optimal patient populations for upcoming trials based on demographic shifts and disease prevalence patterns.
Personalized KAM approaches will emerge through advanced data segmentation. Instead of treating all sites equally, segment accounts by performance tier, therapeutic expertise, and partnership potential. Create customized engagement strategies: high-performing sites get premium support and early access to new trials, while developing sites receive targeted training and resource allocation. Use data insights to match specific site capabilities with appropriate trial protocols.
To build sustainable competitive advantage, implement continuous data-driven optimization:
- Establish a monthly review cadence to analyze account performance metrics
- Create a dashboard tracking site activation times, patient enrollment rates, and protocol compliance
- Use A/B testing for different engagement approaches (digital vs. in-person, frequency of contact)
- Develop a feedback loop where site insights inform protocol design improvements
- Monitor competitor site relationships through public trial registries and investigator publications
Practical tip: Start small by identifying 3-5 key metrics that matter most to your trial success, then build your data infrastructure around those. The most successful organizations will be those that can adapt their KAM strategies in real-time based on emerging data patterns rather than annual planning cycles.
Integrating Competitor Intelligence into KAM Strategy
In today's competitive landscape, understanding your competitors' moves is as crucial as managing your own accounts. Data-driven KAM extends beyond internal metrics to include external market intelligence. By tracking competitor activities, you can anticipate shifts, identify opportunities, and refine your strategy.
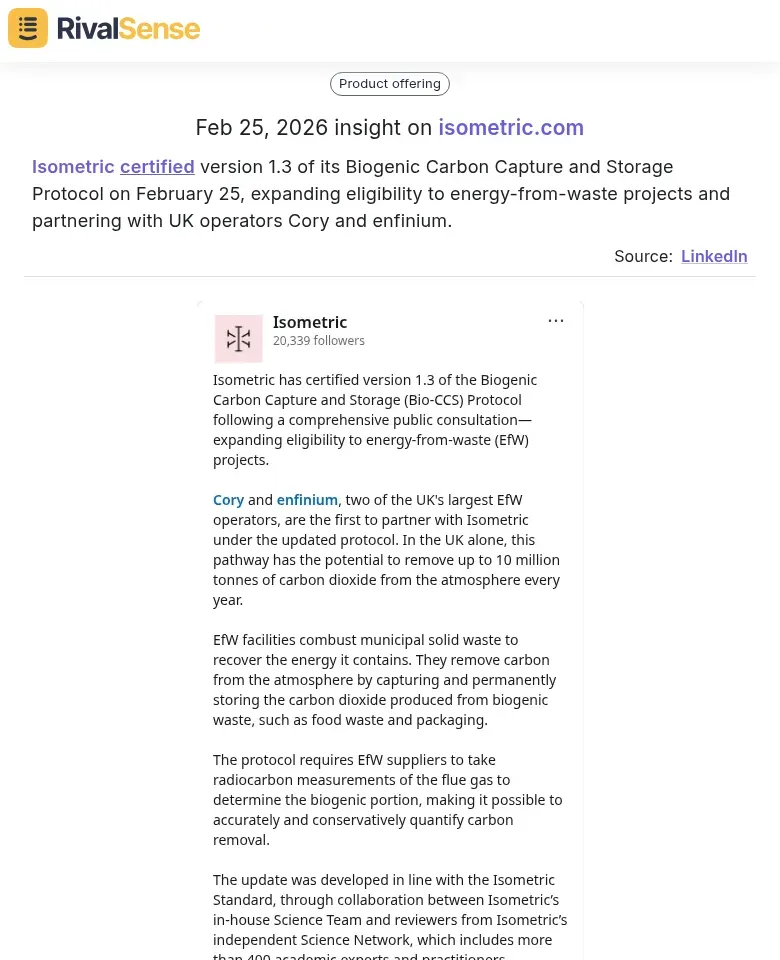
For instance, monitoring product launches and updates can reveal innovation trends. Consider how Isometric certified version 1.3 of its Biogenic Carbon Capture and Storage Protocol on February 25, expanding eligibility to energy-from-waste projects and partnering with UK operators Cory and enfinium. This type of insight is valuable for business strategy as it shows competitor expansion into new areas, allowing you to assess potential threats or partnerships.
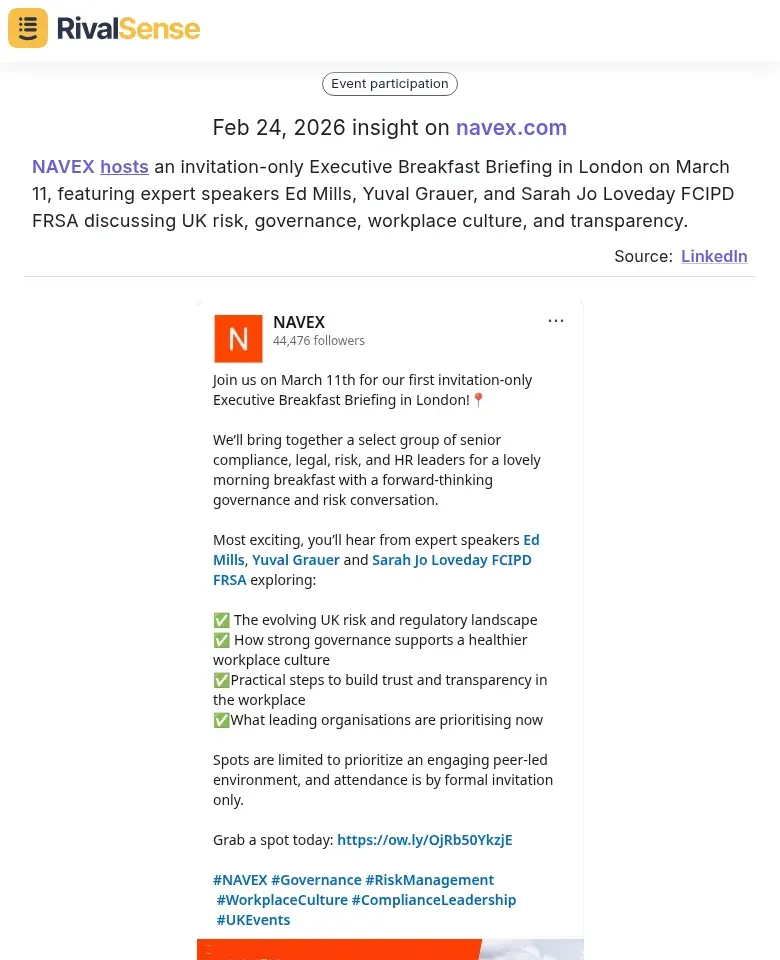
Similarly, tracking event participations provides insights into competitor networking and thought leadership efforts. NAVEX hosts an invitation-only Executive Breakfast Briefing in London on March 11, featuring expert speakers Ed Mills, Yuval Grauer, and Sarah Jo Loveday FCIPD FRSA discussing UK risk, governance, workplace culture, and transparency. Knowing such events helps you understand competitor focus areas and plan your own engagement strategies.
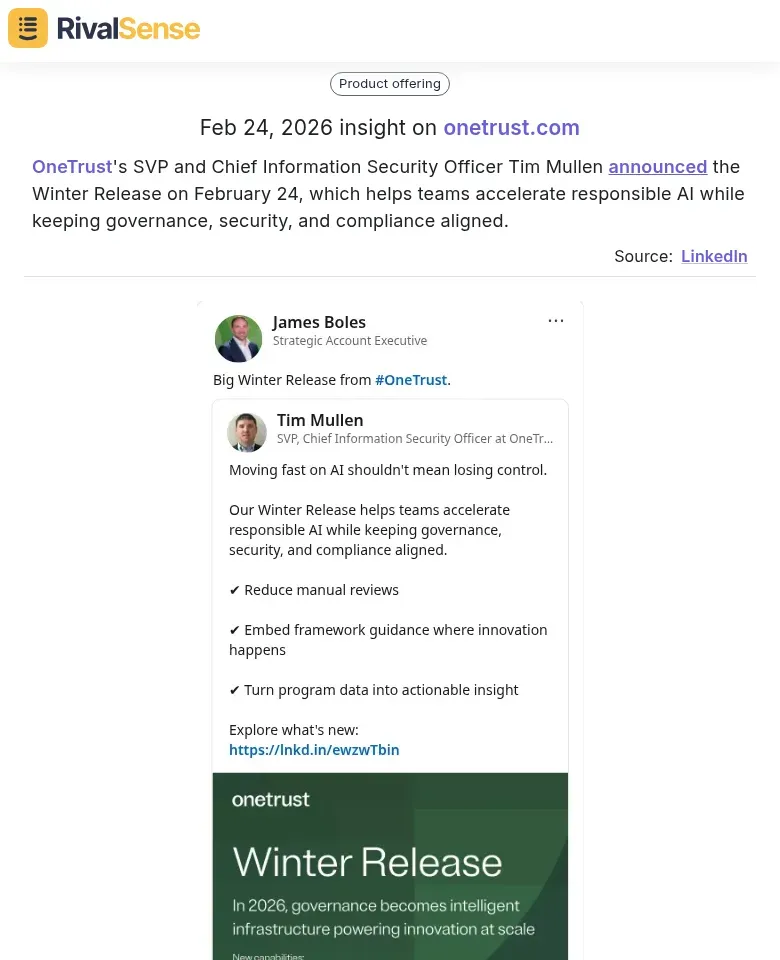
Finally, keeping an eye on product announcements and management changes can signal strategic shifts. OneTrust's SVP and Chief Information Security Officer Tim Mullen announced the Winter Release on February 24, which helps teams accelerate responsible AI while keeping governance, security, and compliance aligned. This insight is valuable for benchmarking your own product roadmap and staying competitive.
Tools like RivalSense automate this tracking, delivering weekly reports on competitor product launches, pricing updates, event participations, partnerships, regulatory aspects, management changes, and media mentions across company websites, social media, and various registries. Incorporating such intelligence into your KAM framework ensures you're not just reactive but proactively shaping your partnerships based on market dynamics.
To assist with the challenges described in this post, try out RivalSense for free at https://rivalsense.co/. Get your first competitor report today and start leveraging competitor intelligence in your KAM strategy.
📚 Read more
👉 How a RivalSense Alert on Jane Street's Lawsuit Delivered Competitive Edge
👉 Twitter Competitor Tracking: The Ultimate Checklist for Key Account Engagement
👉 Pro Audio Key Account Management Cheat Sheet: Best Practices
👉 Avoid These Media Mentions & Content Marketing Competitive Intelligence Mistakes
👉 How to Analyze Competitor Events: A Telenor MWC 2026 Case Study
